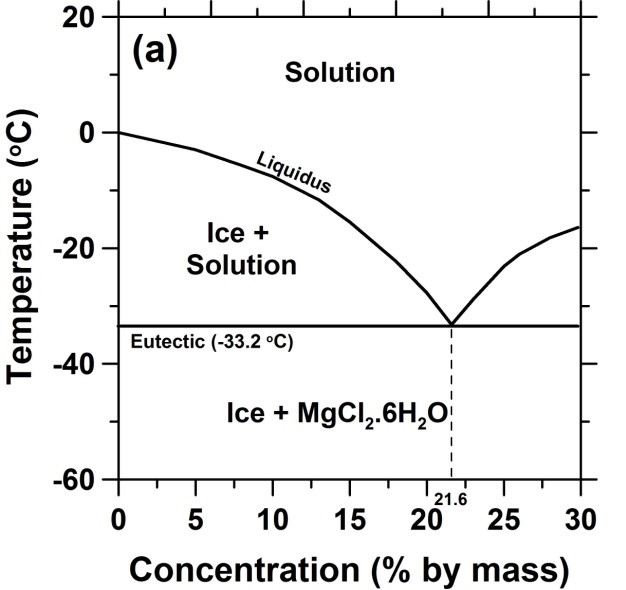 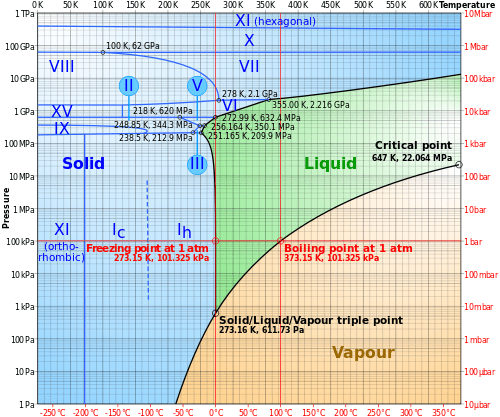
Based on the BET calculations, a list of new eutectic temperatures and compositions of salt hydrate mixtures is given. Salt-water phase diagram of the ternary system (NaCl + NaBO2 + H2O) is significant in separate boron resources from brine. Good agreement with the experimental data was found, when the presumptions of the BET model in respect to water activity and chemical interactions are satisfied. Applying the multi-component formulation of the BET model according to Ally and Braunstein, phase diagrams of aqueous ternary systems composed of the salts LiNO 3, NaNO 3, Mg(NO 3 ) 2, Ca(NO 3 ) 2, Zn(NO 3 ) 2, LiCl, CaCl 2, LiClO 4, and Ca(ClO 4 ) 2 were predicted and compared with available experimental data. The solid phases are ice and the nonahydrate, hexahydrate, tetrahydrate and dihydrate of iron bromide. The parameters were used to model the solid-liquid phase diagrams and to extract the temperature functions of the Gibbs energy of formation of all occurring solid phases with respect to pure liquid water and anhydrous molten salts as reference states. The phase diagram covers the temperature range from minus 40 to 140 ☌. 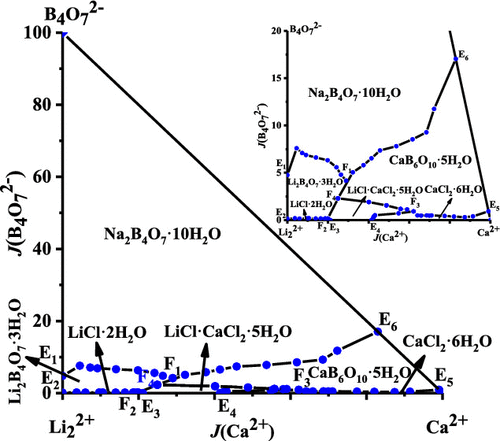
From water activity data within the concentration range of molten salt hydrates the parameters r i and ε i of the modified Brunauer, Emmett and Teller (BET) equation have been determined for several salts i.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
June 2023
Categories |
 RSS Feed
RSS Feed
